Cancer immunotherapy PV-10’s evolution into a cancer immunotherapy
Post Short-Form
We believe that Provectus’ PV-10 (rose bengal sodium or RBS) is a cancer immunotherapy.
PV-10 is a small molecule drug product candidate delivered by intralesional (IL) injection into solid tumors on or inside (via percutaneous injection) the body.
To date, Provectus has generated clinical and preclinical data on PV-10 for the treatment of:
Cutaneous melanoma,
Non-melanoma skin cancers (NMSCs),
Primary liver cancer: hepatocellular carcinoma (HCC),
Cancers metastatic to the liver (e.g., colorectal, uveal, pancreatic),
Breast cancer, and
Potentially (i.e., preclinical) head and neck squamous cell carcinoma (HNSCC).
We believe that PV-10 has the promise of being transformative as a medicine for the prospective traits and characteristics of (i) Provectus’ modernized RBS molecule, (ii) the Company’s proprietary, patented, pharmaceutical-grade RBS drug substance and active pharmaceutical ingredient (API), and (iii) Provectus’ investigational drug product PV-10:
Safety,
Broad-spectrum efficacy,
Wound healing capacity,
Precise, personalized, permanent immune response (Key),
Synergy with standard of care (SOC) medicines, and
Global affordability and accessibility.
Post Long-Form
Introduction
In a world of increasingly complex, expensive, at times incremental, biologics-based cancer therapies with questionable side effects that often have to be managed, we believe that RBS – Provectus’ simply-constructed, synthetic, lead small molecule (of a class of molecules called halogenated xanthenes) – enables PV-10 to (a) possess an exquisite, straightforward, three-stage (i.e., disease treatment, innate immunity, and adaptive immunity), multi-faceted-at-each-stage, situationally-dependent mechanism of action that (b) may treat injectable solid tumor cancers in a fulsome, inter-related, interlocked, systemic manner.
We believe that PV-10 is more than a tool for priming (i.e., stimulating) the immune system for drugs that act against checkpoint proteins, such as CTLA-4, PD-1, or PD-L1 (i.e., immune checkpoint blockade).
We believe that PV-10 has the potential to be a broad-spectrum cancer immunotherapy.
PV-10’s 3-Stage Mechanism of Action and Immune Action
PV-10’s three-stage mechanism comprises:
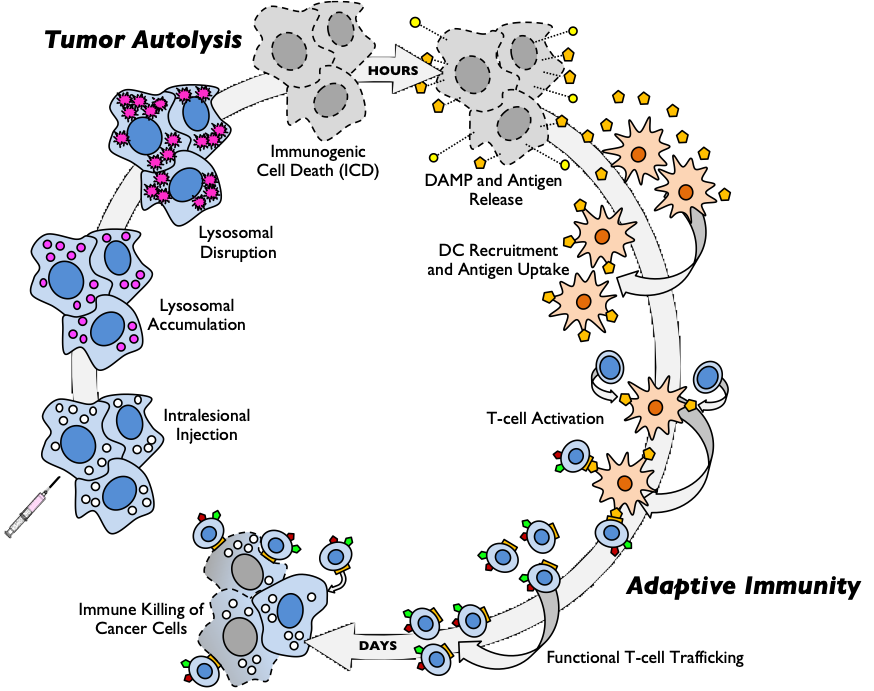
Disease Treatment: The rapid internalization of RBS by tumor cells that disrupts lysosomal integrity, leading to acute autolysis of tumor cells via immunogenic cell death (ICD) that is evident after a single PV-10 injection into a patient’s tumor and occurs within hours of injection,
Innate Immunity: The release of damage associated molecular pattern molecules (DAMPs) and tumor antigens from PV-10-injected tumor tissue that enhances recruitment of dendritic cells (DCs), and
Adaptive Immunity: The priming and activation of a functional T-cell response, including activated CD8+ T-cells, CD4+ T-cells, and NKT cells, that occurs within days of tumor injection by PV-10.
Figure 1. PV-10 Immuno-Oncology Cycle
There is more going on with PV-10’s multi-stage, multi-faceted mechanism than described above and illustrated in Figure 1 (an homage to Chen and Mellman 2013’s original “Cancer Immunity Cycle”). We are creating a new cycle image to better represent what PV-10 may do.
Simply put: “All” that PV-10 cancer immunotherapy does is kill injected tumors rapidly, typically completely, and properly, with one or a few injections. The patient, and their immune system, takes care of the rest.
PV-10: Lysosomal Targeting; Independent, Differential, Temporal Cancer Cell Killing; Stimulatory and Inhibitory
PV-10 (RBS) has a demonstrated, reproducible, replicable, direct mechanism of lysosomal targeting:
We also know, among other things, that:
2011: RBS can trigger at least four different cell death pathways, whose independent temporal activation ensures cancer cell killing when one or several of the pathways are inactivated,
2019: PV-10-enabled cancer cell death is concentration-, time-, and cell-line-dependent, and
2022: PV-10 targets multiple protein kinase signaling pathways, including the phosphorylation of WNK lysine deficient kinase 1 (WNK1). WNK1 potentially regulates important oncogenic pathways, including Wnt signaling by controlling β-catenin levels.
The 2022 poster presentation by Aru Narendran, MD, PhD and his lab team from the Cumming School at the University of Calgary in Alberta, Canada at the annual meeting of the American Association for Cancer Research (AACR) demonstrated an inhibitory trait of the RBS molecule and PV-10 cancer immunotherapy. PV-10 may be considered both stimulatory and inhibitory:
Keytruda® (pembrolizumab) and Opdivo® (nivolumab) inhibit the PD-1 immune checkpoint, and
Oncolytic viruses (OVs), such as Amgen’s Imlygic® (talimogene laherparepvec) and Replimune’s RP1, are stimulatory.
Simply put: PV-10 does not have a monolithic target to kill cancer, kills cancer in different ways depending on the cancer, and kills cancer in different ways if one or other ways are blocked by cancer.
PV-10 is Specific to Cancer Tumors; Keytruda is Not
PV-10’s path to recognized as a cancer immunotherapy started with Provectus and its academic medical research collaborators demonstrating that cancerous tumors injected with PV-10 rapidly and substantially shrunk or destroyed while untreated tumors specific to the PV-10-injected ones also shrunk or were destroyed.
In other words, untreated tumors related to the injected one(s) shrank/made to go away but nothing happened to unrelated untreated tumors:
2012: Melanoma, Moffitt Cancer Center (Moffitt),
2012: Melanoma and hepatocellular carcinoma (HCC), Provectus, and
2013: Melanoma and breast cancer, Moffitt.
PV-10 is a tumor-specific cancer immunotherapy. The contents of the libraries of PV-10-injected tumors are conveyed to the immune system (i.e., to the adaptive immune system via the innate immune system).
PV-10 and Keytruda: PV-10 can complement and be synergistic with checkpoint agents like Keytruda (clinically: cutaneous melanoma and metastatic uveal melanoma; preclinically: HNSCC) and the combination of Yervoy® (ipilimumab) and Opdivo (clinically: metastatic uveal melanoma).
This synergy is exemplified by the natures of PV-10 and checkpoint treatments: PV-10 is a tumor-specific cancer immunotherapy, while Keytruda and Opdivo are non-specific cancer immunotherapies.
You may have read or heard that Keytruda and other checkpoint inhibitors take the brakes off the immune system. Analogies can go too far; however: we suppose that it is okay to release the brakes when the road ahead is straight and flat, but what happens when the road twists and turns, or goes up and down? Keytruda and other checkpoint inhibitors work for some solid tumor cancers, but not for all of them. PV-10 may demonstrate activity in any cancer.
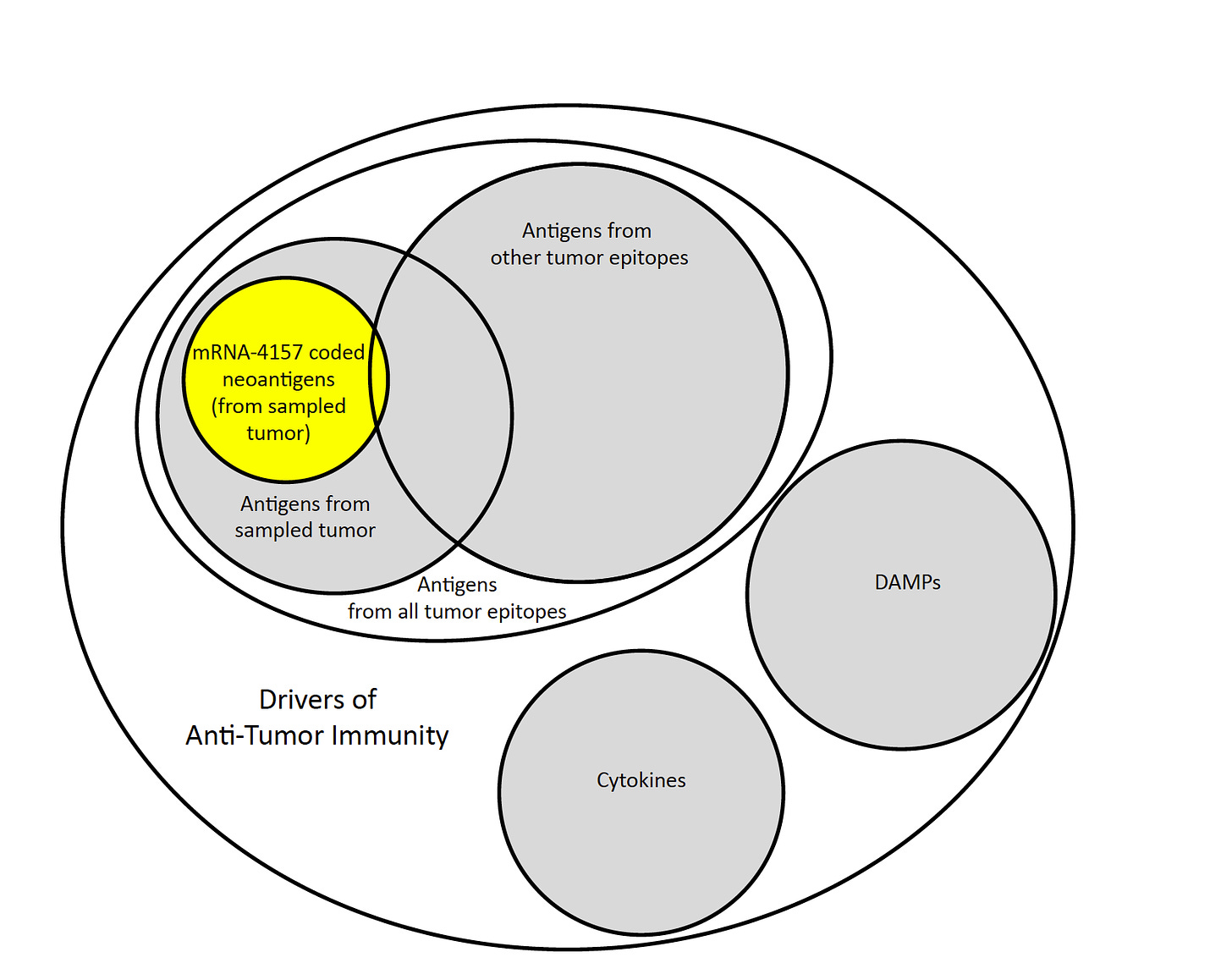
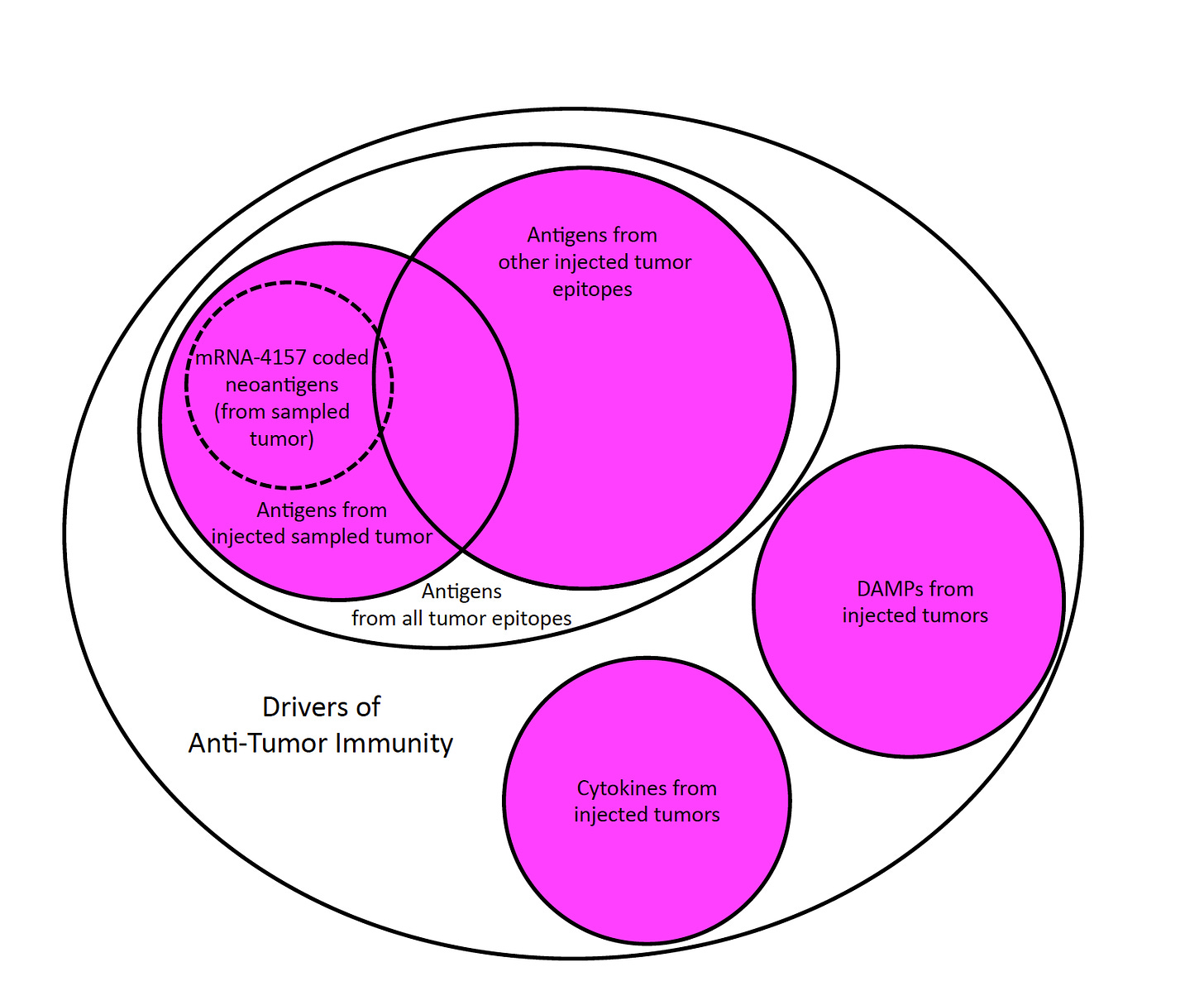
PV-10 vs Moderna’s cancer vaccine mRNA-4157: PV-10’s libraries of injected-tumor contents are the patient’s own libraries (i.e., each PV-10-injected tumor may be a library of germane information for the immune system), while Moderna’s library is their preferred one:
mRNA-4157 produces antigens from a defined library. PV-10 produces the antigens present in each injected tumor of the patient (i.e., the patient’s libraries), which should extend beyond what is contained in Moderna’s library,
mRNA-4157 presumably is based on genomic profiling of a single tumor. PV-10 produces antigens specific to each injected tumor. Moderna or BioNTech will eventually try to sample and profile more tumors, which will be more costly, time consuming, and complex with each additional Moderna- or BioNTech-selected tumor, compared to the simplicity of just injecting a patient’s tumor(s) with PV-10,
mRNA-4157 produces antigens, but presumably does not produce co-stimulatory factors. PV-10 produces antigens and multiple co-stimulatory factors, such as DAMPs, and
mRNA-4157 produces antigenic material without the context of the tumor microenvironment (TME), because Moderna’s vaccine is injected intramuscularly, and not into the tumor itself. PV-10 produces antigens from within the TME because PV-10 is injected into the TME itself.
Figure 2. mRNA-4157 Induces Adaptive Immunity via Production of a Subset of Tumor Antigens from a Sampled Tumor
Figure 3. PV-10 Induces Adaptive Immunity via Synchronous Release of Tumor Antigens from All Injected Tumor Epitopes and Up-Regulatory Signaling Molecules
Cancer Immunotherapy PV-10’s Journey of Self-Discovery
Notably, the above 2012-2013 Provectus/collaborator research and data were presented and published after Provectus completed its Phase 2 clinical trial of monotherapy PV-10 for Stage III and IV melanoma (c. 2010), and data were presented (2009-2014) and published (2014)
And after data from a number of patients with cancers of the liver in Provectus’ Phase 1 liver cancer basket study (i.e., HCC, colorectal [mCRC], non-small cell lung [NSCLC], melanoma, ovarian) was generated and evidence of patients still alive up to nearly five years-to-more than five years upon medical conference presentation was revealed (2015-2017).
Illustratively:
2009: Melanoma presentation,
2014: Melanoma publication, and
2017: Liver publication.
Designing, prioritizing, and executing one’s clinical development program and associated clinical trials thusly demands a real understanding of the practicalities, potentialities, possibilities of one’s medical science and drug product candidate, and, critically, identification of possible gaps in the science and shortcomings of the investigational drug.
Provectus’ prior clinical development program’s approach did not accommodate or integrate the knowledge, wisdom, and, most importantly, earnest belief of RBS’ and PV-10’s immunotherapeutic traits and characteristics into the design of the Company’s initial clinical trials for cutaneous melanoma and cancers of the liver.
We believe that these design principles should include:
Treatment regimen
Principle A: Optimized PV-10 pharmacokinetics.
Clinical management
Principle B: A well-understood, clearly defined patient population,
Principle C: Study endpoints unequivocally supportive of a clearly defined trajectory towards potential, eventual, drug approval, and
Principle D: Well-established, well-reasoned, functional endpoint measurement.
Immuno-narrative
Principle E: Comprehensive immune correlative assessment undeniably demonstrating immunotherapeutic outcome.
Competitive landscape
Principle F: A compelling clinical-business rationale that sufficiently addresses SOC and potential future regulatory approval and/or commercial development.
Putting Phase One of Provectus’ Initial Clinical Development Program into Context
Our 360o approach to analyzing, further exploring, and better understanding PV-10’s authentic immunotherapeutic nature underscored our assessment of Provectus’ initial clinical development program, which we inherited subsequent to a “group” of stockholders (i.e., PRH) entering into a Definitive Financing with the Company in 2017:
Study #1, c. 2007-2010: Provectus’ single-agent PV-10 Phase 2 for Stage III/IV melanoma (NCT00521053)
It transgressed:
Principle C: no measurement of durable response, or survival beyond 12 months, and
Principle D: use of modified RECIST.
Consistent demonstration of (a) a high complete response (CR) rate of PV-10-injected lesions from limited intervention (i.e., 1-2 injetions) and (b) anecdotal demonstration of response in untreated visceral metastases (i.e., brain, lung) were achieved.
Data: 2015 Publication.
Study #2, c. 2009-2015: The Company’s single-agent PV-10 Phase 1 basket study of cancers of the liver (NCT00986661), excluding uveal melanoma work that began in 2018
It transgressed:
Principle A: one injection of PV-10 was administered to one liver tumor of a patient, once, and
Principle C: use of modified RECIST and 2D-EASL.
Consistent demonstration of (a) lengthy OS across multiple tumor types and (b) anecdotal demonstration of response in untreated extra-hepatic disease from limited PV-10 injection were achieved.
In our 2020 presentation, HCC had a median overall survival (OS) of not reached (range 0-112 months) and metastatic colorectal cancer had a median 26.8 months OS (range 3-97+).
Data: 2015 presentation. 2017 presentation (pre-PRH). 2020 presentation (post-PRH).
Study #3, c. 2015-2018: We terminated Provectus’ single-agent PV-10 pivotal Phase 3 trial (NCT02288897)
It transgressed:
Principle B: the trial’s indication of locally advanced cutaneous melanoma (LACM) was not recognized in the medical community; rather, in-transit melanoma (ITM), an artificial sub-population of the LACM construct, is,
Principle D: an inability to treat new lesions (i.e., only baseline disease was injected with PV-10) and an insufficiently long enough period of time prior to address new disease (and also live within a “RECIST world”) without treatment discontinuation for such new lesions, and
Principle F: the cost-benefit was substantially negative, including an addressable market for ITM that is small (i.e., incidence and prevalence are very low).
Consistent demonstration of a high CR rate of PV-10-injected lesions from limited intervention with the Phase 2 trial was achieved. Trial design lessons, good and bad, were learned from Amgen’s (Biovex’s) pivotal single-agent Phase 3 trial for intralesional Imlygic, which was approved in 2015 (publication from 2019).
Data: CT.gov 2022.
Study #4, c. 2015-date: Provectus’ first sponsored combination therapy work, a Phase 1b study of PV-10 and Keytruda for checkpoint-naïve Stage IV melanoma (NCT02557321 main cohort)
It transgressed:
Principle A: five cycles of PV-10 only, and
Principle D: no ability to treat new lesions (i.e., only baseline disease was treated).
We wanted to increase PV-10 treatment; however, the study began enrolling a couple of years prior to PRH. We elected not to demand this protocol change so as not to muddy the analysis and interpretation of the clinical data from the trial. The entire leadership team (old and new) believes, with the benefit of hindsight of course, that overall patient CR and objective response rate (ORR; CR + partial response [PR]), progression-free survival (PFS), and OS rates (from 12-month to 5-year) could have been much higher by (a) optimizing PV-10 treatment as (x) an initial number of cycles of PV-10 followed by (y) treat as needed (pro re nata or PRN), and (b) treating/retreating both baseline and new disease.
Competitive-to-superior overall patient CR (10%), ORR (67%), median PFS (mPFS; 9.7 months), and median OS (mOS; median not reached; 12-month OS rate 95%, 2-year OS rate 62%; 5-year pending) compared pragmatically to other combination therapies and single-agent Keytruda were achieved (given the notable caveat of Provectus’ single-arm trial [SAT]).
Data: 2019 presentation. 2020 presentation.
Study #5, c. 2017-date: Provectus’ single-agent PV-10 Phase 1 study of neuroendocrine tumors (NET) metastatic to the liver (mNET) (NCT02693067)
It transgressed:
Principle A: one injection of PV-10 was administered to one liver tumor of each patient, once, for two-thirds of trial patients, and
Principle D: The study utilized RECIST; however, Provectus did not fully collect PERCIST data to determine if any metabolic complete responses (mCRs) were achieved.
The first mNET patient was treated during the first week of PRH’s leadership at Provectus. Here too, due to the timing of PRH’s transition into the Company, we did not want to change the clinical trial protocol without a then-fuller understanding of the clinical development program (for both melanoma and cancers of the liver). Single-agent PV-10 achieved 83% disease control rate (DCR; disease stabilized, but only PET CT/PERCIST could provide a proper picture of inert diease), 9.2 months mPFS (range 1.0-41.8), and in 2022, 22.5 months mOS (range 5.5-42.3 months, 4 of 12 patients alive), in a heavily pre-treated, third-line patient population. On the immune story side, there was upregulation of NK cells and activated CD4+ T cells in peripheral blood collected 7-28 days post-PV-10 injection. Chromogranin A, a NET marker, was stable post-PV-10 treatment for all patients with stable disease (i.e., 10 of 12 patients).
Data: 2018 presentation. 2021 presentation. 2022 oral presentation.
Study #6: Provectus’ continued combination therapy work, the Phase 1b study of PV-10 and Keytruda for checkpoint-refractory Stage IV melanoma (NCT02557321 expansion cohort 1; c. 2018-date)
It transgressed:
Principle A: Five cycles of PV-10 only, and
Principle D: An inability to treat new lesions within the given time prior to RECIST assessment.
Based on our assessment of the main cohort of this study, we knew that we had to increase PV-10 treatment, particularly because checkpoint-refractory cutaneous melanoma tumors are or become “cold” after failing PD-1 or CTLA-4+PD-1 treatment; however, we again elected not to demand this protocol change because we wanted to reasonably compare and contrast the two cohorts keeping as many degrees of freedom as possible fixed.
Competitive-to-superior overall patient CR (5%), ORR (36%), DCR (64%), mPFS (4.9 months), and mOS (34.1 months, new landmark survival pending) to other combination therapies were achieved (again given the caveat of this Provectus SAT cohort).
We believe that overall patient CR, ORR, mPFS, and mOS could have been much higher with the approach described above of PV-10 PRN. We believe that this was very-preliminarily demonstrated when a non-responder was provided further PV-10 under expanded access following disease progression and trial dropout.
Data: 2020 presentation. 2020 oral presentation. 2021 presentation.
Study #7, c. 2018-date: Provectus’ single-agent PV-10 Phase 1 liver cancer basket study (NCT00986661) expanded to uveal melanoma at a single site (Sapna Patel, MD at MD Anderson Cancer Center)
It evolved in several ways – initially, one injection of single-agent PV-10 was administered to one liver tumor once, we subsequently pushed for more lesions to be treated and more cycles of PV-10 to be given – but transgressed:
Principle D: While the study utilized RECIST and 2D EASL for PV-10-injected lesion response, we did not collect sufficient PERCIST data; however, what PERCIST data we did gather during the course of the study to date enabled us to identify that mCRs were achieved (in one patient who received single-agent PV-10 and three patients who received the combination of PV-10 and checkpoint inhibition).
PV-10-led treatment achieved mCR in 8 of 59 PV-10-injected tumors, 10.7 months mOS in all patients, and mOS was not reached in patients who achieved mCR overall (and who were all stage M1a; disease staging varies from M1a to M1c).
Further, mOS of M1a patients who received PV-10+Yervoy+Opdivo was 44.4 months (caveat: small number of patients). Pragmatically compare this to an mOS of 13.9 months for M1a patients who received Yervoy+Opdivo in a study led by Dr. Patel (Pelster et al. 2021).
Data: 2018 presentation. 2019 presentation. 2022 presentation. 2022 oral presentation A. 2022 oral presentation B.
Study #8, c. 2021-date: Provectus’ further combination therapy work, a Phase 1b study of PV-10 and Keytruda for checkpoint-naïve Stage III melanoma (NCT02557321 expansion cohort 2; 2 of 6 patients came from the main cohort because they were designated as having Stage III disease)
It achieved extraordinary response, PFS, and OS performance to date because we amended this cohort’s clinical trial protocol to, for now, provide PV-10 PRN.
The combination of PV-10 and Keytruda exceeded historical performance of single-agent Keytruda for a similar or the same patient population, albeit with a small number (6) of patients: overall patient CR of 50% and ORR of 83%, mPFS was not reached, and mOS was not reached (18-36 months of study follow-up). Given these results, we plan to continue survival follow-up but do not recruit further for this study cohort.
Data: 2022 oral presentation.
Evolved Clinical Trial Design Principles
We learned a considerable amount about PV-10 and its true immunotherapeutic potential over the course of running Provectus’ Phase One clinical development program through to fruition:
Principle A
PV-10 PRN, which has been and will be incorporated into the clinical trial protocol of any new Provectus or investigator-initiated study, and
Treat/retreat as much baseline and new lesions, if any (see Principle D below).
Principle B
See Provectus’ potential new clinical trials below,
Single-agent PV-10 may be able to have a distinct and influential impact on untreated metastatic disease, and
Target solid tumor cancers where mOS is low and, for now, checkpoint inhibitors don’t work in first-line but may be second-line.
Principle C
CR and mCR, in context (i.e., CR for generally-speaking superficial [i.e., cutaneous or subcutaneous] disease, mCR for visceral disease), may be prognostic of OS, and thus could be prognostic of PFS (to thoughtfully contribute to an evolving regulatory discussion).
Principle D
Treat/retreat as much baseline and new lesions, if any, prior to first RECIST measurement,
Elongate the time period prior to first assessment, but that is acceptable to RECIST, and
For liver cancers, measure response using PERCIST to determine if mCR has been achieved, and to correlate RECIST ORR (i.e., CR or PR) and PFS outcome with potential PERCIST mCR outcome.
Principle E
PV-10 facilitates innate signaling via DAMP release (an emerging story), and
PV-10 activates a precise, specific adaptive response via a function T cell response.
Principle F
See Provectus’ potential new clinical trials below.
Phase Two of Provectus’ New Clinical Development Program
Provectus’ potential next set of clinical trials could comprise one or more of, in no particular order yet:
Study A: The combination of PV-10 and SOC checkpoint for checkpoint-naïve Stage III cutaneous melanoma in a randomized controlled trial (RCT) setting
This potential study derives from the knowledge of Study #8 above.
Prospective Endpoints
Primary: Overall patient CR (RECIST), and
Secondary: Overall patient ORR and PFS (RECIST), and OS.
Clinical rationale
The non-specific checkpoint inhibitor boosts the tumor-specific signal from PV-10 treatment,
First-line setting, and
The clinical trial protocol for this prospective study is effectively complete.
Business rationale
No combination therapy of a checkpoint inhibitor (e.g., Keytruda) and a drug partner has beaten single-agent checkpoint (dun dun duuuun!!!),
If successful, potentially opens the door to co-development with leading checkpoints or one of several me-too approved or investigational checkpoints,
A shorter endpoint timeframe of durable CR (i.e., > 6 months) that leverages PV-10’s effect size in the combination therapy may suggest a smaller patient population and, thus, a swifter interim assessment (i.e., say about 6 months after last patient in), potentially incurring lower financial expense to achieve a potential market catalyst, and
If successful, study biostatistics may provide the foundation for a prospective pivotal Phase 3 trial.
Risks
PV-10 loses, and
If successful, one could still argue about whether PV-10 is doing any, some, or most of the “immune heavy lifting” or Keytruda/the checkpoint is doing all of the work (because some may not be able to wrap their heads and minds around an intratumoral agent being capable of mounting a systemic response to deliver systemic benefit).
Study B: The combination of PV-10 and immunotherapy or dual checkpoints for treatment-naïve, M1a-staged, uveal melanoma metastatic to the liver
This potential study derives from the knowledge of Study #7 above.
Prospective Endpoints
Primary: Overall patient CR & ORR (RECIST) and mCR (PERCIST), and
Secondary: PFS (RECIST) and OS.
Clinical rationale
The non-specific dual checkpoint inhibitor combination (i.e., BMS’ Yervoy+Opdivo) boosts the tumor-specific signal from PV-10 treatment,
Potential first-line setting, pending analysis of M1a patients receiving Kimmtrak® (tebentafusp) in Nathan et al. 2021 (we successfully did this comparison with Pelster et al.), and
The clinical trial protocol for this prospective study is ostensibly complete.
Business rationale
Here too, we are comparing PV-10-led combination therapy to dual checkpoints,
If successful, study biostatistics may provide the foundation for a prospective Phase 2 RCT, and
The other owner of a CTLA-4+PD-L1 combination is AstraZeneca: Imjudo® (tremelimumab) and Imfinzi® (durvalumab).
Risks
PV-10 loses, and
If successful, here too, the question potentially remains of whether PV-10 is doing any, some, or most of the “immune heavy lifting” or is the Yervoy/Opdivo combination doing all of the work.
Study C: The combination of PV-10 and poly-chemotherapy for FOLFIRINOX-refractory pancreatic cancer metastatic to the liver
This potential study derives from the knowledge of a patient with pancreatic ductal adenocarcinoma (PDAC) who received first-line single-agent PV-10 in Study #2 above: one injection to one of two hepatic tumors. The patient did not receive further other treatment and survived for 29 months. For comparison (Conroy et al. 2011), mOS for first-line FOLFIRINOX was 11.1 months and, at 30 months, the OS rate survival was approximately 2%.
Figure 4. PDAC patient treated from Study #2 with a single injection of PV-10 to one hepatic tumor
Prospective Endpoints
Primary: Safety and tolerability; OS, and
Secondary: Overall patient CR & ORR and PFS (RECIST), mCR (PERCIST).
Clinical rationale
PV-10+gemcitabine (gem)+Nab-paclitaxel (nab),
Second-line setting,
While gem+nab in a first-line setting achieved an mOS of 8.5 months (Von Hoff et al. 2013), second-line gem+nab refractory to FOLFIRINOX had an mOS of 6.6 months (Huffman et al. 2023), and
The clinical trial protocol for this prospective study is effectively complete.
Business rationale
We think that chemotherapy is not considered an immunotherapy (some may hypothesize that chemotherapy may have immunotherapeutic properties (e.g., inhibition of myeloid suppressor cells [MDSCs], macrophages),
Thus, we believe that if the combination of PV-10+gem+nab is successful, most folks should intellectually honestly conclude that PV-10 did some, most, or substantially all of the “immune heavy lifting,” thus potentially establishing PV-10 in most eyes as a cancer immunotherapy,
Keytruda was granted approval in 2017 (accelerated) and 2023 (full) for patients with unresectable or metastatic microsatellite instability–high (MSI-H) or mismatch repair–deficient (dMMR) solid tumors, including pancreatic cancer, and
Our goal for this study would be to exceed the mOS of second-line gem+nab (to “win” the study at hand when compared to historical SOC mOS) and mOS for first-line FOLFIRINOX (since the clock for OS starts ticking for a patient upon initial treatment, and potentially then on to other lines of therapy).
We have discussed internally the prospects of a pancreatic study as early as 2018, having worked with translational researchers at Moffitt (Shari Pilon-Thomas, PhD and her lab team) who published germane preclinical research in 2021 (this research was presented in stages at the annual meetings of the Society for Immunotherapy of Cancer [SITC] in 2016 and 2020), and a prospective principal investigator in the disease area since 2019:
The entire leadership team has wanted to do a properly-designed study in metastatic pancreatic cancer, due to, but not limited to, the terrible nature of the disease itself, having turned down a number of expanded access requests over the years, and knowing directly and indirectly that Provectus was not design-ready (for the many reasons enumerated and principles described above), and
In many respects, PV-10’s achievement of multiple mUM mCRs in Study #7 above significantly changed things (such as providing us much more confidence in PV-10), together with the firming of Principles A through F.
Risk
PV-10 loses.
4. Still in draft form
Provectus disclosed in its Form 10-Q for the period ending March 31, 2023 filed with the SEC last Thursday that PV-10 (referenced as ITU PV-10, or intratumoral PV-10, in the 10-Q filing) “…has undergone and is undergoing preclinical monotherapy and combination therapy study for the treatment of…human papillomavirus-positive and negative [HPV+/-] head and neck squamous cell carcinoma at Moffitt.”
This HPV+/- HNSCC research, which started in 2019, was and is pro bono. We have only supplied PV-10 to the researchers. Further exploration will be done, and we hope that summary preclinical data may be accepted for AACR 2024.
We believe that we have a very preliminary framework for a prospective clinical study, which could include comparing PV-10 to Opdivo or chemotherapy in a second-line setting.
The clinical rationale for PV-10 in this setting should be similar to that of Provectus’ work in cutaneous melanoma: rapid destruction of PV-10-injected lesions with limited intervention, a systemic, tumor-specific immune response, and, in particular, injection-site wound healing.
Figure 4. A Melanoma Patient from Provectus’ Phase 1 Clinical Trial Receiving PV-10
The business rationale for PV-10 in this second-line setting could be potentially, finally, in a design-ready manner, prioritizing taking on a checkpoint (i.e., Opdivo) head-to-head, rather than initially chemotherapy.
Where to Find Available Information
Investors and others should note that Provectus Biopharmaceuticals, Inc. (the Company) uses, and will continue to use, press releases distributed by GlobeNewswire®, filings with the Securities and Exchange Commission (SEC), and the Company's website (provectusbio.com) to disclose material financial and operational information to the Company's shareholders, prospective investors, the media, and others interested in the Company. The Company also intends to use certain social media accounts as a means of disclosing information and observations about the Company and its business, and for complying with the Company's disclosure obligations under Regulation FD: the Provectus Substack account (provectus.substack.com), the @ProvectusBio Twitter account (twitter.com/provectusbio), and the Company's LinkedIn account (linkedin.com/company/provectus-biopharmaceuticals). The information and observations that the Company posts through these social media channels may be deemed material. Accordingly, investors should monitor these social media channels in addition to following the Company's press releases, SEC filings, and website. The social media channels that the Company intends to use as a means of disclosing the information described above may be updated from time to time.
Forward-Looking Statements
The information in Provectus’s Substack post may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in Provectus’s Substack are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the Securities and Exchange Commission, including those described in Item 1A of:
the Company’s Annual Report on Form 10-K for the year ended December 31, 2022, and
Provectus’ Quarterly Report on Form 10-Q for the period ended March 31, 2023.