Cancer Immunotherapy PV-10's Innate Immune Signaling
DAMPs and related cytokines have immune up-/down-regulatory roles in cancer, potentially indicative of disease burden, immune competence, and therapeutic effect
We previously mentioned in Provectus’s Substack post Cancer immunotherapy PV-10’s evolution into a cancer immunotherapy that we were working on a new PV-10’s immuno-oncology (IO) cycle. This new IO cycle, which displays small molecule-based PV-10’s mechanism of action and dual innate-adaptive mechanisms of immune action, is illustrated in Figure 1 below.
Substack Post Figure 1. New PV-10 IO Cycle
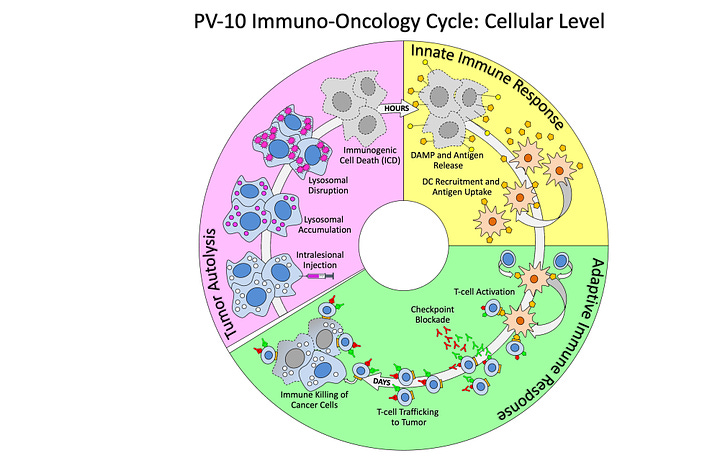
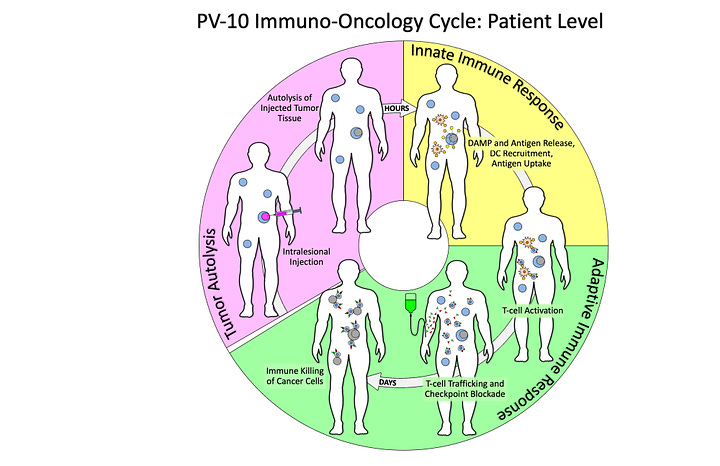
This new PV-10 IO cycle representation:
Adds a patient-level view of PV-10’s mechanisms (right image) to the original cellular-level mechanistic view (left image),
Now highlights the innate immune response resulting from PV-10 injection of tumors, and
Maintains the involvement of immune checkpoint blockade (i.e., checkpoint inhibition) for medical and business rationales.
We specifically note that PV-10’s three-step, interconnected, systemic mechanisms of action and immune action necessarily involves an innate immune response directly related to PV-10 treatment:
Tumor tissue cell death in PV-10-injected tumors within hours,
Innate immune signaling from the release of damage-associated molecular pattern molecules (DAMPs), tumor antigens, cytokines, etc. from PV-10-injected tumors, and
A tumor-specific functional T cell response within days of PV-10 injection.
As a reminder, our PV-10 IO cycle draws from the original 2013 IO cycle of Chen and Mellman, “Oncology Meets Immunology: The Cancer-Immunity Cycle” (Immunity. Volume 39, Issue 1, 25 July 2013, Pages 1-10). For its great communicative strengths, one deficit of the Chen and Mellman concept is the very notion of an IO cycle when applied to drug treatments (see Chen and Mellman’s Figure 3 below).
Substack Post Figure 2. Chen and Mellman 2013’s Figure 3, Therapies that Might Affect the Cancer-Immunity Cycle
PV-10’s IO cycle starts with PV-10’s injection of a patient’s cancer tumors and ends with the actions of the patient’s innate and adaptive immune systems, which is why our IO cycle has a sliver of white space between the Tumor Autolysis and Adaptive Immune Response segments. That is, PV-10’s IO “cycle” is not a cycle.
You will also note in our new PV-10 IO cycle (Figure 1 of this Substack post) that we include the involvement of immune checkpoint blockade; see the green intravenous bag in the third Adaptive Immune Response segment: an “homage” to Keytruda® (pembrolizumab) and its green-colored branding.
PV-10 is a tumor-specific cancer immunotherapy. The contents of the libraries of PV-10-injected tumors are conveyed to the immune system (i.e., to the adaptive immune system via the innate immune system). PV-10 can complement and be synergistic with checkpoint agents:
+Keytruda: cutaneous melanoma and metastatic uveal melanoma (mUM) (Provectus clinical data), head and neck squamous cell carcinoma (HNSCC) (Provectus pre-clinical), and
+the combination of Yervoy® (ipilimumab) and Opdivo® (nivolumab): mUM (Provectus clinical).
This synergy between PV-10 and checkpoint blockade is exemplified by the respective natures of PV-10 and checkpoint treatments: PV-10 is a tumor-specific cancer immunotherapy, while Keytruda and Opdivo are non-specific cancer immunotherapies. The joint mechanism of immune action of PV-10/checkpoint combination therapy can be described as “induce-and-boost:” Keytruda or Yervoy+Opdivo boost what PV-10 induces; that is, the checkpoint or checkpoints boost PV-10’s tumor-specific immune response.
Ongoing Analysis of Clinical DAMPs Data
DAMPs and related cytokines can have both immune up-regulatory and down-regulatory roles in cancer, and their pattern and levels in peripheral blood can be indicative of disease burden, immune competence, and therapeutic effect.
Peripheral blood collected from mUM patients in Provectus’s liver cancer basket study (NCT00986661) are being analyzed for DAMPs (e.g., HMGB1, HSP90a, S100A8, S100A9, S100A12, S100B, IL-1a, etc.). We are currently probing, among other things but not limited to, relationships such as baseline DAMPs versus outcome (overall survival or OS) or versus disease burden, change in DAMPs versus outcome (OS) or versus PV-10 dose, and chronology of change in DAMPs.
There is a reasonable body of literature describing the complex role of DAMPs in cancer:
HMGB1
While high mobility group box 1 (HMGB1) has been identified as playing a role in initiation of PV-10-induced immune activation [Liu et al., 2016; Innamarato et al., 2021], it can also be prognostic for outcome based on its role in promoting accumulation and immunosuppressive function of myeloid-derived suppressor cells (MDSCs) [Kang et al., 2013; Parker et al., 2014], and chronic elevation may compromise anti-tumor immunity. A clinical review [Fucikova et al., 2015] noted that HMGB1 over-expression has been shown to correlate with advanced clinical stage or decreased disease-free and/or overall survival in pa@ents with bladder carcinoma, nasopharyngeal carcinoma, colorectal cancer (CRC), hepatocellular carcinoma (HCC), HNSCC, and prostate cancer; and that elevations of HMGB1 in serum have been correlated with incidence, progression or unfavorable disease outcome in pancreatic adenocarcinoma, laryngeal squamous cell carcinoma, and malignant pleural mesothelioma; conversely, a treatment-related increase in the circulating levels of HGMB1 has been associated with pathological complete response or partial remission amongst breast carcinoma patients treated with neo-adjuvant chemotherapy based on epirubicin (an immunogenic cell death {ICD} inducer), as well as amongst subjects with chemotherapy-refractory tumors treated with oncolytic virus. Cell culture work on uveal melanoma cells identified a correlation between HMGB1 expression and invasiveness of uveal melanoma (UM) cells, suggesting potential as a biomarker [Pardo et al., 2006], but there appear to be no reports validating this in patients.
HSP90
Heat shock protein 90 (HSP90) is highly expressed by many tumors, and plays a central role in key signaling pathways controlling melanoma development, progression and response to therapy. An early study of tumor specimens showed that HSP90 expression was significantly higher in tumors than nevi and was associated with disease progression, identifying HSP90 as a possible biomarker and therapeutic target [McCarthy et al., 2008], and it has been a target for inhibition in a number of subsequent therapeu@c efforts [MielczarekLewandowska et al, 2020]. A phase 2 study of an HSP90 inhibitor (ganetespib) in pa@ents with mUM showed modest clinical benefit and rapid development of resistance [Shah et al., Mel Res 2018]. HSP90a has been reported to promote conversion of monocytes into MDSCs, and was associated with shortened progression-free survival (PFS) in cutaneous melanoma patients [Arkhypov et al., 2022].
S100A8 and S100A9
S100A8 and S100A9 have been implicated in the development and spread of lung cancer [Ehrchen et al., 2009], and S100A4 has been shown to correlate with the progression of non-small cell lung cancer (NSCLC) [Chen et al., 2008]. S100A8/A9 were shown to be elevated in serum and play an up-regulatory role in growth and metastasis of a murine colon tumor model [Ichikawa et al., 2011]. In vitro and in vivo studies of S100A8/A9 showed cell growth-promoting activity at low concentration and an important role in breast cancer invasion/metastasis [Yin et al, 2013]. Evaluation of serum samples from breast cancer pa@ents and healthy volunteers identified elevation of S100A9 as a potential diagnostic marker, whereas S100A12 levels were equivalent in both groups [Gunaldi et al., 2015]. In bladder cancer patients, elevation of S100A12 in plasma has been identified as a biomarker [Elamin et al., 2020]. Expression of S100A9 and S100A12 were found to be significantly higher in patients with prostate cancer metastases compared to patients without clinically detectable metastases. And S100A8 and S100A9 were shown to activate mitogen-activated protein kinase (MAPK) and nuclear factor kappa B (NF-κB) signaling in pancreatic cancer, thereby inducing metastatic spread [Nedjadi et al., 2018]. Early data on S100 proteins in cutaneous melanoma identified elevated serum S100 as a biomarker of advanced metastasis [Henze et al., 1997; Jury et al., 2000]; subsequent work identified S100B as the specific biomarker, which has been correlated with tumor mass and survival, with decreasing S100B concentrations reflecting response to therapy and increasing concentrations indicating progression [Harpio and Einarsson, 2004]. A small study of 18 mUM patients identified a correlation between serum S100B and metastasis to the liver, and an approximate 3-fold elevation was observed after onset of metastasis in 8 pa@ents with pre- and post-metastasis serum samples [Barak et al., 2007]. This was further supported in a follow-on report on 28 mUM pa@ents, 16 of whom (57%) exhibited elevated S100B vs a control group of 43 non-metastatic uveal melanoma pa@ents [Barak et al., 2011; Hendler et al., 2011]. However, a larger recent study of 101 mUM patients showed low frequency of elevation of S100B at first diagnosis of Stage IV disease (20.7%), while only half of pa@ents (54%) exhibited rising serum S100B levels during disease progression; the authors concluded that serum S100B is not a reliable marker for early detection of advanced UM [Salzmann et al., 2023].
IL-1a
The cytokine IL-1a has been implicated in increased metastatic potential of pancreatic cancer [Melisi et al., 2009; Xu et al., 2010; Hernandez et al., 2016]. Recent investigation in a B16 mouse cutaneous melanoma model showed that IL-1a can serve an innate pro-inflammatory role and that immunotherapy-induced IL-1a can increase resistance to further immunotherapy. In this work, tumor size was inversely correlated with response to immunotherapy: large tumors had higher levels of IL-1a, Th2 cytokines, polymorphonuclear myeloid-derived suppressor cells (PMN-MDSCs), and regulatory T cells but lower levels of IL-12, Th1 cytokines, and activated T cells [Singh et al., 2021].
Baseline DAMPs vs Outcome (OS)
Provectus’s current mUM clinical data suggest that one or more of these DAMPs can be prognostic for outcome: transient increases in one or more of these DAMPs following PV-10 administration could play an important role in the initiation anti-tumor immunity, whereas chronic elevation at baseline may indicate poor anti-tumor immune competence and prognosis.
Baseline DAMPs vs Disease Burden
As with OS, HSP90a, S100A8, and HMGB1 exhibited substantial correlation with baseline disease burden, while IL-1a proved to have a particularly strong and significant correlation. These results are consistent with literature reports for each of these DAMPs, supporting their roles as biomarkers of advanced disease and poor prognosis in mUM. Interestingly, while S100B has been proposed as an important biomarker in mUM, Provectus’s correlation data are consistent with recent results from Salzmann et al., 2023, showing minimal prognostic value for this DAMP. A further implication of total metastatic disease burden was that long-surviving subjects, having received PV-10 treatment, tended to have lower metastatic burden (i.e., M1a staging).
Change in DAMPs vs Outcome (OS)
Although no statistically significant changes were observed from this analysis, there was a tendency for several DAMPs (i.e., HSP90a, S100A9, S100A12) to exhibit an increased mean level at day 28. Prior testing for changes in DAMPs following PV-10 treatment have shown significant increases of HMGB1 in samples collected 7-14 days post-treatment of cutaneous melanoma patients, with a decrease in HMGB1 toward baseline levels in samples collected 21-28 days post-treatment [Liu et al., 2016]. This suggests that sampling 28 days after PV-10 administration may substantially miss post-treatment DAMP release.
Early DAMP Release
Data for the subset of participants with samples collected 1 or 7 days post-PV-10 were analyzed: HMGB1, HSP90a, and S100A9 exhibited marked increases at day 1 or 7 versus baseline, and these increases trended toward baseline in day 28 samples. The aforementioned changes in HMGB1, HSP90a, and S100A9 were more pronounced in the high-PV-10 dose group.
Change in DAMPs vs PV-10 Dose
Change from pre-PV-10 treatment to day 28 follow-up was evaluated by bifurcating mUM patients by median PV-10 dose (threshold of 5 mL of PV-10) to assess whether a dose response was evident. A similar analysis was done using using a threshold of 2 mL of PV-10.
Using the 5 mL PV-10 threshold, significant changes in HSP90a and S100A12 were observed in the high-PV-10 dose group, while S100B showed a significant decrease in the low-PV-10 dose group. The 2 mL PV-10 threshold yielded similar results, with increases in S100A12 and IL-1a in the high-dose group. These results suggest that PV-10 dose may play a role in the magnitude of DAMP release.
Preliminary Conclusions
Baseline DAMPs appear to be prognostic for outcome.
DAMP release following PV-10 administration appears to occur in a predictable manner that is consistent with non-clinical models and clinical data of PV-10 from other solid tumor cancer indications. Increased DAMP release in high-PV-10 dose participants implies a dose response.
The collection of samples for DAMP analysis at day 28 remains important, but may reduce sensitivity to DAMP changes from baseline; sample collection 1-14 days post-PV-10 may increase sensitivity to apparent PV-10-induced DAMP release from the injection of hepatic uveal melanoma metastases.