Statistical modeling of PV-10 and chemotherapy for FOLFIRINOX-refractory pancreatic cancer metastatic to the liver
Study PV-10-PA-01 (Phase 1a): 2nd-line therapy of PV-10 + gemcitabine + nab-paclitaxel vs historical 2nd-line SOC of gemcitabine + nab-paclitaxel
At Provectus’s 2023 Annual Stockholder Meeting, we described three potential new or continued studies that could comprise Part Two of Provectus’s Clinical Development Program for PV-10 (see slide no. 19).
One of these prospective clinical trials is a pancreatic cancer study where intralesional (IL) PV-10 is administered to a patient’s hepatic tumors, who also receives the standard of care (SOC) second-line combination therapy of two chemotherapies, gemcitabine (GEM; which has been described as the backbone of the treatment for resectable pancreatic cancer) and nab-paclitaxel (PAC; a new formulation of paclitaxel free of solvents) (GEM-PAC).
Together with working closely with the prospective principal investigator (PI) of the prospective lead clinical trial site, our preparatory work prior to study launch includes, but is not limited to:
defining the trials’s patient population,
finalizing the study’s design,
budgeting and financial planning,
working through regulatory strategy and affairs, and
identifying and assessing potential study milestones for data readouts.
This study is comprised of two parts:
a dose escalation portion of 6 patients (i.e., Phase 1a), and
a dose expansion portion of up to 26 patients (i.e., Phase 1b).
This Provectus Substack post discusses our statistical modeling work, which is a part of our study design discussion and debate with our PI and during our internal meeting sessions.
The Biology of Metastasis in Pancreatic Cancer
The liver is the principal site of metastasis for pancreatic cancer:
An expansive survey of epidemiology data on 13,233 metastatic pancreatic cancer patients in the NCI SEER database (2010-2013) by Oweira et al. 2017 showed that the liver was the most common site of metastasis (76% of patients), with the lungs a distant second (20%), followed by lymph nodes (9%), bone (7%), and brain (1%).1 The authors note that another important site of metastasis (the peritoneum) is not included in the SEER database, and hence was not included in their tabulation.
A meta-analysis using source data from two large first-line studies conducted by Wu et al. 2021 showed that 84% of 640 patients with metastatic pancreatic cancer had liver metastases, 50% had lymph node involvement, 26% had lung metastases, and 22% had metastatic disease of the peritoneum.2
In the first-line setting, a pivotal study of FOLFIRINOX vs GEM by Conroy et al. 2011 showed that 88% of 341 patients with metastatic pancreatic cancer had liver metastases, 26% had lymph node involvement, 24% had lung metastases, and 19% had metastatic disease of the peritoneum.3
A similar first-line pivotal study of GEM-PAC vs GEM monotherapy by Von Hoff et al. 2013 showed that 84% of 861 patients with metastatic pancreatic cancer had liver metastases, 39% had lung metastases, and 3% had metastatic disease of the peritoneum (nodal status was not reported).4
The Conroy and Von Hoff studies demonstrated survival benefits that established
FOLFIRINOX as first-line and GEM-PAC as second-line SOC for metastatic pancreatic cancer.5
A review of 10 studies of GEM-PAC second-line to FOLFIRINOX in advanced or metastatic pancreatic cancer showed a similar metastatic pattern, where among 552 metastatic patients for whom the site of metastasis was reported, 84% patients had liver metastases, while 41% had peritoneal disease, 25% had lung metastases, and 23% had lymph node involvement.6
The liver is also a clinically important site of metastasis:
Oweira et al. 2017 concluded that patients with isolated liver metastases have a
significantly worse survival prognosis compared to those with isolated lung or distant nodal metastases. Interestingly, the authors found no difference in the outcome for patients with single sites of metastasis vs those with multiple metastatic sites.
Analyzing the data from the Van Hoff study, Tabernero et al. 2015 found that the
presence of liver disease was strongly prognostic for a negative outcome: patients with liver metastases had a median overall survival (mOS) of 8.3 months vs 11.0 months for those without liver disease (hazard ratio of 1.75 by a multivariate Cox model, HR = 1.81 by stepwise multivariate analysis, P < 0.001 for both models).7 The number of metastatic sites was also prognostic for survival, with 2 or more sites presenting a consistently negative prognosis.
The Wu et al. 2021 meta-analysis found a similar trend, where patients with isolated liver or peritoneal metastases had a much poorer overall survival (OS) than those with isolated lung metastases. In line with Tabernero, number of metastatic sites was also shown to be an independent prognostic factor for OS, although approximately two-thirds of patients had only one or two sites (i.e., 34% and 37% of patients, respectively).
Survival Data in the Second-Line Setting
Historical survival data for GEM-PAC as second-line to FOLFIRINOX in metastatic pancreatic cancer are available from a number of sources. The fraction of patients with metastatic disease (vs locally advanced Stage III disease) and, among metastatic patients, the fraction with hepatic metastasis, are variable. Studies where at least 80% of patients had metastatic disease include:
Portal et al. 2015:8
mOS = 8.8 months (CI 6.2-9.7 months, N = 57)
All patients had metastatic disease (72% had liver metastasis)
Zhang et al. 2015:9
mOS = 5.3 months (CI NR, N = 28)
82% had metastatic disease (metastatic site NOS)
Mita et al. 2019:10
mOS = 7.6 months (CI 5.7-8.6 months, N = 30)
80% had metastatic disease (67% had liver metastasis)
Huh et al. 2021:11
mOS = 9.9 months (CI 7.5-12.4 months, N = 40)
88% had metastatic disease (55% had liver metastasis)
King et al. 2022:12
mOS = 6.6 months (CI 6.2-7.1 months, N = 1,281)
All patients had metastatic disease (NOS - not otherwise specified)
Zaitbet et al. 2022:13
mOS = 7.1 months (CI NR, N = 219)
All patients had metastatic disease (71% had liver metastasis)
Huffman et al. 2023:14
mOS = 6.6 months (CI 4.7-8.4 months, N = 40)
92% had metastatic disease (70% had liver metastasis)
The weighted average mOS for these studies is 6.8 months (range 5.3-9.9). Because King et al. 2022 represents far more patients than the other studies, an alternate, normalized weighted average was calculated by de-emphasizing King by a factor of 10; this yielded an es4mated mOS of 7.3 months. Note that the longest mOS in this group was reported by Huh et al. 2021 for a cohort of patients the smallest fraction having hepatic disease.
Several other studies included a larger fraction of patients with locally advanced disease:
Nguyen et al. 2017:15
mOS = 12.4 months (CI NR, N = 30)
Patients were second-line to FOLFIRINOX
62% had liver metastasis (23% had Stage III disease)
Palacio et al. 2018:16
mOS = 5.2 months (CI 4.9-11 months, N = 25)
Patients were second-line or greater without prior GEM (no further history provided)
Chae et al. 2020:17
mOS = 9.8 months (CI 8.9-10.6 months, N = 102)
Patients were second-line to FOLFIRINOX
35% had liver metastasis (25% had Stage III disease)
The weighted average mOS for these studies is 9.6 months (range 5.2-12.4), illustrating the marked impact inclusion of Stage III patients can have on survival of the study population in historical control data sets.
Statistical Modeling using Historical Controls
To evaluate the relevance of mOS for a small cohort, such as that proposed for the Phase 1a portion of Provectus’s potential study PV-10-PA-01, the statistical modeling software G*Power 3.1 was used with the following assumptions to calculate the sample size for a typical phase 1 (80% power) single-arm study of an investigational treatment (H1; i.e., PV-10 + GEM-PAC) vs a defined control (H0; i.e., GEM-PAC):
Mean H0 (mOS historical control): 7.3 months
Mean H1 (mOS PV-10 treatment arm): 10.3 months
SD: 2.5 months
𝛼 error probability: 0.05
Power (1-𝛽 error probability): 0.80
Calculated sample size: 6
This shows that a sample size of six patients can exhibit meaningful preliminary differences (i.e., at 80% power) against the available historical control data. These calculations are illustrated below.
The data in King et al. 2022 illustrate the potential relevance of landmark OS data relative to historical controls (Figure 5(a), OS from commencement of second-line standard combination chemotherapy, i.e., GEM-PAC), where 1-year and 2-year OS are approximately 23% and 8%, respectively.

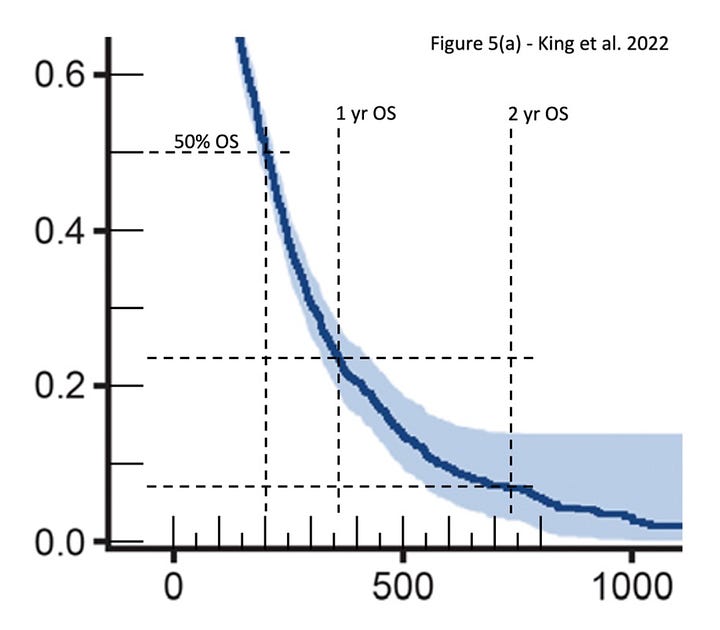
Conclusions
Hepatic metastases are common in metastatic pancreatic cancer and are associated with increased mortality. Although the exact mechanism of this poor prognosis is unknown, it suggests that the control of liver disease may improve clinical outcome. Intralesional PV-10 combined with GEM-PAC may be a promising approach to control the liver disease of metastatic pancreatic cancer, directly addressing this risky hepatic disease burden while improving immune recognition of non-PV-10-injected disease beyond the site(s) of IL PV-10 administration.
As a preliminary gauge of the relevance of this approach, any deviation from the modeling criteria described here (mOS and landmark OS) will facilitate the assessment of the success or failure of the proposed investigational treatment based on the most clinically-meaningful outcome in the proposed patient population.
It is notable that the historical control data contain a sizable fraction of patients without hepatic disease and, as such, represent an upper limit on survival for the proposed PA-01 population, in which 100% of patients will have hepatic disease.
Oweira H, Petrausch U, Helbling D, Schmidt J, Mannhart M, Mehrabi A, Schob O, Giryes A, Decker M, Abdel-Rahman O. Prognostic value of site-specific metastases in pancreatic adenocarcinoma: A Surveillance Epidemiology and End Results database analysis. World J Gastroenterol. 2017 Mar 14;23(10):1872-1880. https://doi.og/10.3748/wjg.v23.i10.1872.
Wu L, Zhu L, Xu K, Zhou S, Zhou Y, Zhang T, Hang J, Zee BC. Clinical significance of site-specific metastases in pancreatic cancer: a study based on both clinical trial and real-world data. J Cancer. 2021 Jan 18;12(6):1715-1721. https://doi.org/10.7150/jca.50317.
Conroy T, Desseigne F, Ychou M, Bouch. O, Guimbaud R, Becouarn Y, Adenis A, Raoul JL, Gourgou-Bourgade S, de la Fouchardiere C, Bennouna J, Bachet JB, Khemissa-Akouz F, P.r.-Verg. D, Delbaldo C, Assenat E, Chauffert B, Michel P, Montoto-Grillot C, Ducreux M; Groupe Tumeurs DigesGves of Unicancer; PRODIGE Intergroup. FOLFIRINOX versus gemcitabine for metastaGc pancreaGc cancer. N Engl J Med. 2011 May 12;364(19):1817-25. https://doi.org/10.1056/NEJMoa1011923.
Von Hoff DD, Ervin T, Arena FP, Chiorean EG, Infante J, Moore M, Seay T, Tjulandin SA, Ma WW, Saleh MN, Harris M, Reni M, Dowden S, Laheru D, Bahary N, Ramanathan RK, Tabernero J, Hidalgo M, Goldstein D, Van Cutsem E, Wei X, Iglesias J, Renschler MF. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013 Oct 31;369(18):1691-703. https://doi.org/10.1056/NEJMoa1304369.
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines), Pancreatic Adenocarcinoma, version 2.2023, Jun 19, 2023.
Review conducted by Provectus, source data cited infra.
Tabernero J, Chiorean EG, Infante JR, Hingorani SR, Ganju V, Weekes C, Scheithauer W, Ramanathan RK, Goldstein D, Penenberg DN, Romano A, Ferrara S, Von Hoff DD. Prognostic factors of survival in a randomized phase III trial (MPACT) of weekly nab-paclitaxel plus gemcitabine versus gemcitabine alone in patients with metastatic pancreatic cancer. Oncologist. 2015 Feb;20(2):143-50. https://doi.org/10.1634/theoncologist.2014-0394.
Portal A, Pernot S, Tougeron D, Arbaud C, Bidault AT, de la Fouchardiere C, Hammel P, Lecomte T, Dreanic J, Coriat R, Bachet JB, Dubreuil O, Marthey L, Dahan L, Tchoundjeu B, Locher C, Lep.re C, Bonnetain F, Taieb J. Nab-paclitaxel plus gemcitabine for metastatic pancreatic adenocarcinoma amer Folfirinox failure: an AGEO prospective multicentre cohort. Br J Cancer. 2015 Sep 29;113(7):989-95. https://doi.org/10.1038/bjc.2015.328.
Zhang Y, Hochster H, Stein S, Lacy J. Gemcitabine plus nab-paclitaxel for advanced pancreatic cancer after first-line FOLFIRINOX: single institution retrospective review of efficacy and toxicity. Exp Hematol Oncol. 2015 Oct 7;4:29. https://doi.org/10.1186/s40164-015-0025-y.
Mita N, Iwashita T, Uemura S, Yoshida K, Iwasa Y, Ando N, Iwata K, Okuno M, Mukai T, Shimizu M. Second-Line Gemcitabine Plus Nab-Paclitaxel for PaGents with Unresectable Advanced Pancreatic Cancer after First-Line FOLFIRINOX Failure. J Clin Med. 2019 May 29;8(6):761. https://doi.org/10.3390/jcm8060761.
Huh G, Lee HS, Choi JH, Lee SH, Paik WH, Ryu JK, Kim YT, Bang S, Lee ES. Gemcitabine plus Nab-paclitaxel as a second-line treatment following FOLFIRINOX failure in advanced pancreatic cancer: a multicenter, single-arm, open-label, phase 2 trial. Ther Adv Med Oncol. 2021 Nov 10;13:17588359211056179. https://doi.org/10.1177/17588359211056179.
King G, Ittershagen S, He L, Shen Y, Li F, Villacorta R. Treatment Patterns in US Patients Receiving First-Line and Second-Line Therapy for Metastatic Pancreatic Ductal Adenocarcinoma in the Real World. Adv Ther. 2022 Dec;39(12):5433-5452. https://doi.org/10.1007/s12325-022-02317-9.
Zaibet S, Hautefeuille V, Auclin E, Li.vre A, Tougeron D, Sarabi M, Gilabert M, Wasselin J, Edeline J, Artru P, Bechade D, Morin C, Ducoulombier A, Taieb J, Pernot S. Gemcitabine + Nab-paclitaxel or Gemcitabine alone after FOLFIRINOX failure in patients with metastatic pancreatic adenocarcinoma: a real-world AGEO study. Br J Cancer. 2022 Jun;126(10):1394-1400. https://doi.org/10.1038/s41416-022-01713-w.
Huffman BM, Basu Mallick A, Horick NK, Wang-Gillam A, Hosein PJ, Morse MA, Beg MS, Murphy JE, Mavroukakis S, Zaki A, Schlechter BL, Sanoff H, Manz C, Wolpin BM, Arlen P, Lacy J, Cleary JM. Effect of a MUC5AC Antibody (NPC-1C) Administered With Second-Line Gemcitabine and Nab-Paclitaxel on the Survival of PaGents With Advanced Pancreatic Ductal Adenocarcinoma: A Randomized Clinical Trial. JAMA Netw Open. 2023 Jan 3;6(1):e2249720. hZps://doi.org/10.1001/jamanetworkopen.2022.49720.
Nguyen KT, Kalyan A, Beasley HS, Singhi AD, Sun W, Zeh HJ, Normolle D, Bahary N. Gemcitabine/nab-paclitaxel as second-line therapy following FOLFIRINOX in metastatic/advanced pancreatic cancer-retrospective analysis of response. J Gastrointest Oncol. 2017 Jun;8(3):556-565. https://doi.org/10.21037/jgo.2017.01.23.
Palacio S, Hosein PJ, Reis I, Akunyili II, Ernani V, Pollack T, Macintyre J, Restrepo MH, Merchan JR, Rocha Lima CM. The nab-paclitaxel/gemcitabine regimen for paGents with refractory advanced pancreatic adenocarcinoma. J Gastrointest Oncol. 2018 Feb;9(1):135-139. https://doi.org/10.21037/jgo.2017.10.12.
Chae H, Jeong H, Cheon J, Chon HJ, Ryu H, Kim IH, Kang MJ, Jeong JH, Ryoo BY, Kim KP, Yoo C. Efficacy and safety of second-line nab-paclitaxel plus gemcitabine amer progression on FOLFIRINOX for unresectable or metastatic pancreatic ductal adenocarcinoma: multicenter retrospective analysis. Ther Adv Med Oncol. 2020 May 27;12:1758835920923424. hZps://doi.org/10.1177/1758835920923424.





I hope recruitment can get under way soon.